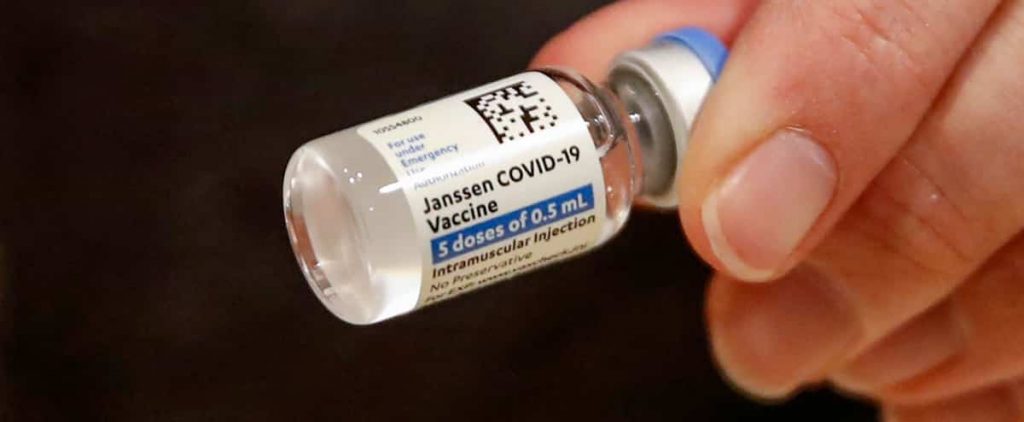
The Johnson & Johnson vaccine label, the first doses expected this week in the country, was updated Monday by Health Canada in order to prevent secondary risks occurring during its injection.
The vaccine package supplement now lists the signs and symptoms of side effects Canadians may experience after receiving the vaccine, as well as when patients need to seek medical attention immediately.
This update was determined after a few cases of blood clots were reported in the United States when a person was given a dose of this vaccine.
“Health Canada has worked closely with the manufacturer and other international regulatory bodies to evaluate the latest evidence and take action before the vaccine is available to Canadians,” the ministry said in a series of tweets posted in the afternoon.
However, Health Canada reiterated that “the benefits of vaccination outweigh these very rare risks” and supported the use of this vaccine, just as it did with AstraZeneca.
Johnson & Johnson vaccine, recommended for people over the age of 18, was licensed on March 5 in Canada.
Prime Minister Justin Trudeau announced Friday that the first doses of the vaccine should be delivered this week, in order for distribution to begin in May.
To see, too

“Music guru. Incurable web practitioner. Thinker. Lifelong zombie junkie. Tv buff. Typical organizer. Evil beer scholar.”






More Stories
Possibility of forgery of 787 documents The US aviation regulator opens an investigation into Boeing
British Columbia protects mobile home residents by amending the law
Losses of $748 million in 2023: Change of course necessary for Canada Post